Bloch Equations, Dipolar Interactions, and Chemical Shifts
Learn how Bloch equations, dipolar interactions, and chemical shifts shape NMR and MRI. Understand spin dynamics, magnetic coupling, and resonance frequency shifts to interpret molecular structure and chemical environments.
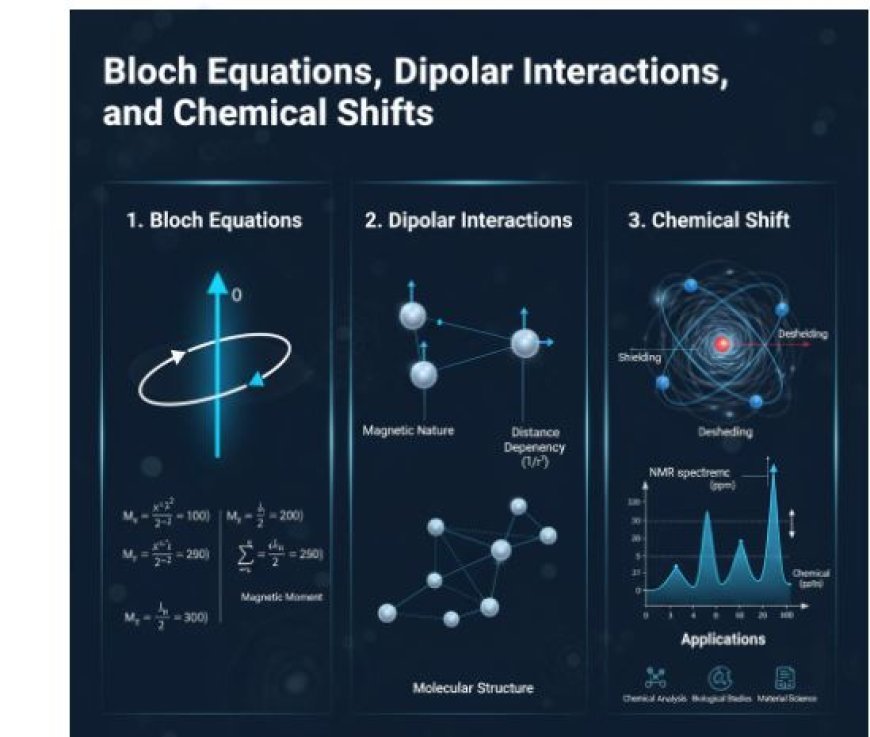
Understanding Bloch Equations, Dipolar Interactions, and Chemical Shifts
- Bloch Equations, Dipolar Interactions, and Chemical Shifts has many interesting things going on, mainly in the areas of nuclear magnetic resonance (NMR) and magnetic resonance imaging (MRI). These things help us understand and study materials.
- Bloch equations, dipolar interactions, and chemical changes are three important ideas in this field.
1. Bloch Equations
- When nuclei are in a magnetic field, the Bloch equations show how the magnetic moments of those nuclei behave.
- For methods like MRI and NMR, they are very important because they give us a way to think about how nuclear spins change when they are exposed to magnetic fields.
A. Important Parts of the Bloch Equations
- For example, the magnetic moment is a vector number that shows how magnetic a centre is. It is changed by the spin of the centre and how it interacts with a magnetic field outside of it.
- Precession: When nuclei are in a magnetic field, their magnetic moments don't just point in the field's direction; they also change directions, like a spinning top wobbling.
B. Mathematical Representation
- There are a number of differential equations that make up the Bloch equations.
- The part of the x-axis (Mx) The part of the y-axis that is (My) The part of the z-axis (Mz).
- These parts explain how the spins change over time when they are affected by a magnetic field and other forces.
C. Physical Meaning
- We can use these equations to figure out how much energy the spins take in or give off during NMR tests. This helps us learn more about the structure and motion of molecules.
2. Interactions Between Dipoles
- The forces that act between two magnetic dipoles, which you can think of as tiny magnets, are called dipolar interactions.
- Nuclear spins communicate with each other in NMR in a very important way through dipolar interactions.
A. Things that make dipolar interactions unique.
- Nature of magnetism: Each spin in a nucleus works like a tiny magnet. Two magnetic moments that are close to each other change the direction of the other moment.
- Dependency on Distance: The dipolar interactions become weaker as the square of the distance between nuclei grows. This means that nuclei that are closer together will connect more strongly than those that are farther apart.
Why it's important in NMR
B. How it relates to NMR
- The lengths and arrangements of atoms in a molecule can be found out by their dipolar interactions. Scientists can use this to figure out the makeup of complicated chemicals.
3. Chemical Shift
- In NMR spectra, chemical shift is an occurrence that tells us a lot about the chemical surroundings of nuclei.
- It refers to the small change in a nucleus's resonant frequency that is caused by the magnetic field that electrons around it create.
A. Causes of Chemical Shifts
- Electronic Environment: The magnetic field a nucleus experiences is affected by nearby electrons in a way that depends on the atoms and functional groups in the molecule. This changes the amount of energy the nucleus needs to flip its magnetic moment.
- Shielding: Nuclei in areas with more electron density (more shielding) will have a lower effective magnetic field, which will cause a lower resonant frequency than those in areas with less shielding.
B. Measuring Chemical Shifts:
Chemical shifts are important for finding functional groups and understanding molecular structures; they are usually measured in parts per million (ppm).
C. Applications in Chemistry Chemical shifts help scientists figure out which bonds or groups are present in a substance. This makes it easier to study proteins, organic compounds, and other materials.
IMAGE SOURCE (THUMBNAIL)
What's Your Reaction?