The Stark Effect in Quantum Mechanics
The Stark Effect refers to the splitting and shifting of atomic or molecular energy levels when subjected to an external electric field. It arises from quantum mechanical interactions between the electric field and the charged particles (mainly electrons) within the atom. The effect can be linear (energy shift proportional to the field strength) or quadratic (shift proportional to the square of the field). Using perturbation theory, the Stark Effect can be mathematically described by how an atom’s dipole moment interacts with the electric field. This phenomenon is observed in hydrogen atoms, hydrogen-like ions, and polar molecules, and is crucial in spectroscopy, quantum information science, plasma diagnostics, laser cooling, and astrophysics.

The Stark Effect
1. Introduction to the Stark Effect
The Stark Effect occurs when an atom or molecule is placed in an external electric field. The charged components of the atom, such as electrons and protons, interact with this field, leading to changes in energy levels.
- Energy Level Shifts and Splitting: When an electric field is applied, it modifies the atom’s energy levels. In many cases, degenerate energy levels (energy levels that originally had the same energy) split into multiple distinct levels.
- Quantum Mechanics: The Stark Effect arises due to quantum mechanical interactions between the electric field and the charged particles in the atom, mainly the electrons.
2. Types of Stark Effect
The Stark Effect can be classified into two main types:
1. Linear Stark Effect:
- This occurs when the energy level shift is directly proportional to the strength of the electric field.
- It is commonly observed in cases where the electric field does not interact strongly with the atom, such as when the energy level is low or when the dipole moment of the atom is small.
2. Quadratic Stark Effect:
- This occurs when the energy level shift is proportional to the square of the electric field strength.
- It is observed in polar molecules or atoms with a permanent dipole moment.
- The energy shift increases with stronger electric fields, leading to a more complex splitting pattern.
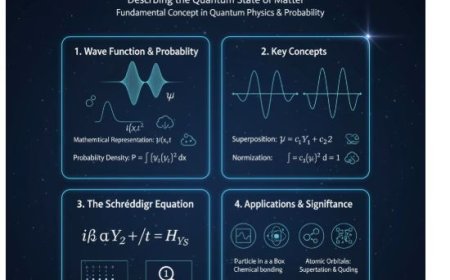
3. Mathematical Explanation
- In quantum mechanics, perturbation theory can be used to describe the energy shifts caused by the Stark Effect.
- The electric field interacts with the atom’s dipole moment, modifying the system’s Hamiltonian (total energy operator).
- For an atom with an electric dipole moment d, the external electric field E affects its Hamiltonian through the interaction term:
The energy shift due to this interaction is given by:
where:
- ψ represents the quantum state of the system.
- d is the electric dipole moment.
- E is the applied electric field.
- This equation describes how the interaction between the dipole moment and the electric field modifies the energy levels of the atom.
4. Physical Explanation and Key Concepts
- Dipole Moment: Atoms or molecules with a permanent dipole moment experience a stronger Stark Effect. The dipole moment measures how separated the positive and negative charges are within an atom or molecule. The larger the dipole moment, the greater the Stark Effect.
- Selection Rules: When an electric field is present, the selection rules for energy level transitions change. Certain transitions that were previously forbidden can occur due to the electric field.
- Degeneracy Lifting: Some energy levels in an atom or molecule are degenerate (they have the same energy) when there is no electric field. When an electric field is applied, this degeneracy is removed, and the levels split into multiple distinct levels.
5. Examples of the Stark Effect
1. Hydrogen Atom:
- A well-known example is the Stark Effect in hydrogen atoms.
- When an electric field is applied, the degenerate energy levels of the hydrogen atom split into multiple levels.
- In weak electric fields, the energy shift is linear with respect to the field strength.
2. Hydrogen-Like Ions:
- Ions similar to hydrogen (single-electron ions) exhibit a similar Stark Effect.
- The presence of an electric field causes the spectral lines of these ions to split and shift.
3. Molecules with a Permanent Dipole Moment:
- The Stark Effect is stronger in molecules with a permanent dipole moment, such as water (H₂O).
- In molecular systems, the effect is more complex, and both linear and quadratic Stark effects can occur simultaneously.
6. Applications of the Stark Effect
1. Spectroscopy:
- The Stark Effect is widely used in atomic and molecular spectroscopy.
- By studying how energy levels shift and split in an electric field, scientists can gain insights into the structure of atoms and molecules.
2. Quantum Information Science:
- The Stark Effect is useful in quantum computing and quantum control.
- Controlling atomic and molecular energy levels with external electric fields helps in the development of quantum bits (qubits) for quantum information processing.
3. Plasma Physics:
- The Stark Effect is used to study plasmas (ionized gases).
- It helps in determining electric fields in high-energy environments such as fusion reactors and astrophysical plasmas.
4. Laser Technology:
- The Stark Effect is applied in laser cooling and trapping.
- It allows for precise control of atomic and molecular energy levels, aiding in manipulating atomic motion.
5. Astrophysics:
- The Stark Effect is observed in the spectra of stars and celestial bodies.
- It helps scientists measure the strength of electric fields in space environments.
What's Your Reaction?