Electronic Spectroscopy of Diatomic Molecules
Learn how electronic spectroscopy and vibrational coarse structure in diatomic molecules reveal electronic–vibrational transitions, molecular dynamics, and spectral patterns useful in chemistry, physics, and material science.
Electronic Spectroscopy of Diatomic Molecules
Vibrational Coarse Structure
Introduction
- Electronic spectroscopy is a powerful technique in physics and chemistry used to study electron transitions in molecules.
- In diatomic molecules, electronic transitions are closely linked to vibrational states.
- Understanding these transitions helps in analyzing molecular behavior and interactions.
1. Introduction to Electronic Spectroscopy
1.1 What is Electronic Spectroscopy?
- Electronic spectroscopy examines interactions between electromagnetic energy and molecular electronic states.
- When a molecule absorbs or emits light, electrons transition between different energy levels.
1.2 Diatomic Molecules
- Diatomic molecules consist of two atoms (either the same element or different elements).
- Their simple structure makes them ideal for spectroscopic studies.
2. The Role of Vibrational States
2.1 Overview of Vibrational States
- In addition to electronic transitions, molecules also undergo vibrational motion.
- These vibrations include:
- Stretching (changes in bond length)
- Bending (changes in bond angle)
2.2 Interaction Between Vibrational and Electronic States
- When a molecule undergoes an electronic transition, it can also change vibrational states.
- This interaction leads to vibrational coarse structure in the electronic spectrum.
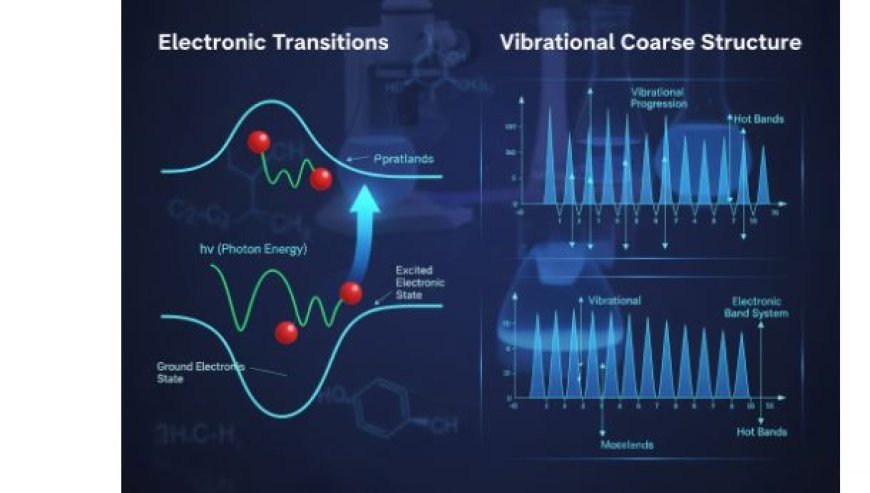
3. Vibrational Coarse Structure
3.1 Definition
- Vibrational coarse structure refers to patterns of spectral lines caused by simultaneous electronic and vibrational transitions.
- These patterns help analyze the vibrational effects in electronic spectra.
3.2 Progressions and Sequences
In vibrational coarse structure, spectral lines appear in specific progressions and sequences.
3.2.1 Progressions
- Definition: Progressions occur when vibrational levels change within the same electronic state.
- Observation: Each spectral line corresponds to a different vibrational transition, and the spacing between them provides information on vibrational energy levels.
3.2.2 Sequences
- Definition: Sequences involve transitions between different vibrational levels in two electronic states.
- Types:
- Hot bands: Occur when transitions start from excited vibrational levels rather than the ground state.
- Electronic band systems: Show vibronic coupling (interaction between vibrational and electronic states).
4. Factors Affecting Vibrational Coarse Structure
4.1 Vibrational Frequencies of Molecules
- Each molecule has unique vibrational frequencies that define its spectral pattern.
- These frequencies depend on:
- Bond strength
- Atomic masses
4.2 Electronic Coupling
- The interaction between electronic states affects vibrational structures.
- Strong vibrational-electronic coupling enhances specific transitions in the spectrum.
4.3 Temperature Effects
- Temperature influences population distribution among different vibrational levels.
- At higher temperatures, hot bands appear more prominently.
5. Applications of Electronic Spectroscopy
5.1 Molecular Identification
- Electronic spectroscopy helps identify molecules by analyzing unique spectral fingerprints.
- Used in fields like:
- Astronomy (studying molecules in space)
- Climate science (analyzing atmospheric gases)
5.2 Understanding Molecular Dynamics
- Helps scientists study molecular motion and reactions.
- Useful for investigating chemical reaction mechanisms and energy transfer processes.
5.3 Material Characterization
- Crucial in materials science and nanotechnology.
- Helps in:
- Understanding electronic properties of materials
- Developing new spectroscopic methods
IMAGE SOURCE (THUMBNAIL)
What's Your Reaction?